Preventing newborn RSV

85.9%
reduction in hospitalizations for infants who received Beyfortus compared to those who did not (2023–24 RSV season)
$1.8B
in first-year sales of Beyfortus

“A big thank you for creating fantastic resources that have made our roles so much easier!”
Worldwide, childhood vaccination hesitancy among parents fluctuates between 25-45%, according to the World Health Organization (WHO), a trend it considers one of the biggest global threats to public health.
Sanofi was the first company to supply the polio vaccine worldwide. Because of the global fight against the disease, endemic wild poliovirus transmission is now limited to just two countries. However, attitudes toward vaccines and immunizations have shifted significantly. To ensure a smooth launch of Beyfortus in a time of increased vaccine hesitancy, Sanofi asked IDEO to conduct research across four important and very different healthcare markets: Germany, Spain, China, and the US.

IDEO worked with parents, caregivers, healthcare professionals, and healthcare administrators to identify potential barriers to vaccine adoption and understand the emotional needs of all stakeholders involved in the immunization process. Navigating cultural differences and diverse healthcare environments, the team uncovered a set of insights.
For healthcare professionals, the team discovered there was no one-size-fits-all approach to immunization training across Sanofi’s 20-plus global markets. While consistency of key information was important, trainings needed to be flexible and customizable. This way, regardless of whether the vaccine was administered by midwives, OB-GYNs, nurses, or doctors, they could confidently discuss the pros and cons across cultural contexts and administer the drug correctly.
For new parents, receiving information about a new immunization immediately after giving birth could be too stressful. But simply leaving pamphlets about Beyfortus in waiting rooms didn't address their real concerns, either. Caregivers needed to engage in conversations earlier in their pregnancies and continue them after leaving the hospital, when they might have additional questions.

IDEO translated these research insights into a highly accessible global communication toolkit that uses simple, straightforward language, metaphors, and illustrations to support informed conversations between healthcare providers and parents before, during, and after a child is born. IDEO then conducted a series of market-fit co-design workshops with representatives of Sanofi’s global immunization business to iterate on the print and digital tools, make them more resonant with regional customers, and ensure compliance with local regulations. IDEO handed the toolkits over for development to support the launch of Beyfortus in January 2024, just in time for RSV season.

Beyfortus saw enormous demand in its first year, demonstrating strong efficacy in real-world applications and achieving blockbuster status as a $1.8 billion therapy. Sanofi’s global launch team credits the patient-centric toolkit with increased requests for communication materials from its regional markets as well as from healthcare and government organizations that have typically been hard to reach, key goals of its global-local launch strategy.
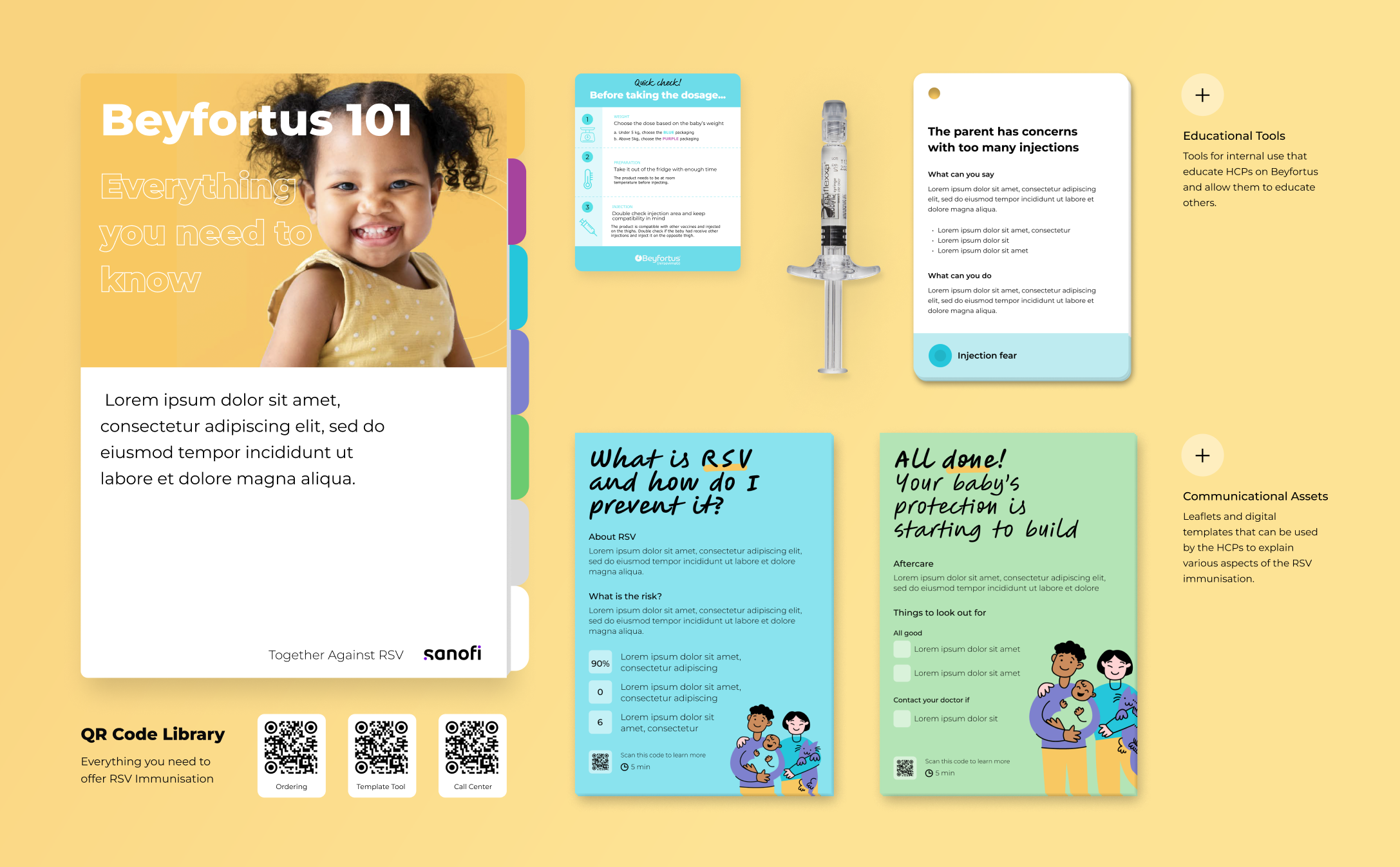
Since its development, Beyfortus has received swift regulatory approval from the US, EU, China, and Japan, with patient demand causing Sanofi to triple its worldwide manufacturing capacity to meet the needs of the 2024-25 RSV season. Ahead of the larger global distribution of Beyfortus, IDEO and Sanofi partnered with equity expert consultants, Fearless Futures, to conduct a DEI review and worked with doctors and nurses who participated in the initial drug launch to make the materials even more accessible, relevant, and successful for 2025 and beyond. To date, six million babies—and counting—have been immunized with Beyfortus.
Curious about how this kind of thinking could benefit your organization? We’d love to hear from you.
Subscribe

.svg)